
The main feature of an elimination reaction in organic chemistry is that two substituents are removed from a molecule, forming a double bond. There are two main types of elimination reactions; E1 and E2. The E1 elimination is a type of elimination reaction in organic chemistry. It takes place in two separate steps, but one of the reactants only limits the reaction rate. The E2 elimination is a type of elimination reaction in organic chemistry. It takes place in only 1 step, but two reactants limit the reaction rate.

Image from Labster's Elimination Reaction: Use cyclohexanol to create polymers Virtual Lab.
Before going for any topic, the basics of the topic must be cleared. To teach the organic compound, you must first clear the concepts of basic organic chemistry. They don't know about the functional groups, attacking and leaving groups in any compound. Before going further, you must clarify what saturated and unsaturated organic compounds are. They also face difficulties when they have to identify any organic compound by the IUPAC name.
In organic chemistry, there is more than one possible reaction and reaction mechanism. Students face difficulties selecting the major pathway for the compounds, so you must be clear when there is more than one functional group, what the major product will be, and which mechanism it will follow. If there is more than one attacking and leaving group, students get confused about which group will attack and which one is the good leaving group. These terms must be cleared first to study any organic compound and its reactions.
Theoretical studies of organic compounds and reactions are tricky because most compounds have 3D structures. Organic reactions involve complex reactions mechanisms that are difficult to understand theoretically. You can use 3D models of organic molecules so that students easily understand the structures of molecules. For reactions, mechanisms must use video graphics; if possible, draw the complete mechanism along with arrows identifying the reaction sites and reacting groups.
Before going to organic reactions following terms must be cleared:
Carbocations: A carbocation is a reactive intermediate that has a carbon atom bearing a positive charge and three bonds to that carbon instead of four.
Alkyl halides: Alkyl halides (also known as haloalkanes) are hydrocarbon compounds in which one or more of the hydrogen atoms have been replaced by a halogen atom (iodine, bromine, chlorine, or fluorine). Incorporating halogen atoms into a hydrocarbon changes the compounds' physical properties - affecting size, electronegativity, bond length, and strength.
Regioselectivity: The term regioselective can be used to describe any process that favors bond formation at a particular atom over other possible atoms. The ‘Regio-’ prefix comes from the Latin for ‘Region’ or an area of something. A reaction has high regioselectivity when one major product dominates due to the reaction being highly favored at one atomic position over another.
Stereochemistry: Stereochemistry relates to the three-dimensional arrangement of atoms and molecules and the effect of this spatial arrangement on chemical reactions.
A leaving group is a part of a molecule that can break away (leave the molecule) during a reaction. The key factor contributing to a species' suitedness as leaving group is its basicity: The weaker the base, the better the leaving group. Halogens are often used as the leaving group, e.g., in alkyl halides. The general order of "ability to leave" for them is I > Br > Cl > F.
A catalyst is a component in a chemical reaction that increases the reaction rate by changing the reaction mechanism. A catalyst is not used in the reaction and thus is often effective in sub-stoichiometric amounts.
A lone pair is a pair of valence electrons that do not take part in bonding.
A substrate is a chemical specie that is consumed during a chemical reaction to give a product.
The main feature of an elimination reaction in organic chemistry is that two substituents are removed from a molecule, forming a double bond. Often, one of the substituents will be hydrogen (H+).
Figure 1. Generic representation of an elimination reaction. Two substituents are removed from the reactant, and the product contains a double bond (Image source Labster theory).
The E1 elimination is a type of elimination reaction in organic chemistry. It takes place in two separate steps, but one of the reactants only limits the reaction rate. Figure 1 & 2 here provides an example of E1 elimination.
1)
The leaving group - bromide - takes the electrons from the detaches from the main molecule, which becomes a carbocation. Carbocations are not stable, so some further reaction must occur!
2)
A base abstracts a proton from the main molecule, which stabilizes the main molecule via the formation of a double bond.
Because the reaction goes through a planar carbocation intermediate, there is no specific selectivity towards cis or trans for the product. Usually, the result will be a mix of the two.
The E2 elimination is a type of elimination reaction in organic chemistry. It takes place in only 1 step, but the rate of the reaction is limited by two of the reactants.
The leaving group takes the electrons, detaches from the main molecule as a base, and abstracts a proton. Notice there is no intermediate formation of a carbocation.
As the E2 reaction takes place in a single step, the reaction products will all have the same configuration, and the product with trans configuration will be far more dominant. This is due to the fact that the leaving group and the base-removed hydrogen will tend to be antiperiplanar, that is, on opposite sides of the bond of the two carbons that the involved groups are connected to. This is a unique trait of the E2 reaction.
These factors generally increase reactivity towards an E1 elimination:
A good leaving group in the main molecule
Degree of substitution: The more substituted the (potential) carbocation is, the more stable and, therefore likely to form it is. E1 almost only occurs at tertiary carbon atoms!
Base: A strong base that can pull off that hydrogen. But E1 can take place even with a weak base.
Temperature: The rate of elimination reactions generally increases with temperature. In competition with substitution reactions, adding heat will favor the elimination path.
The solvent in which the reaction takes place can have a huge effect. How this affects elimination reactions are beyond this simulation, but you can check out this page for an overview of how solvents are normally categorized.
E2 eliminations are for many factors influenced in a similar way as for the E1 type.
Noteworthy differences are:
Degree of substitution: Even though elimination reactions are generally more favored in highly substituted molecules, E2 reactions can also occur with secondary and even primary carbon atoms.
It usually requires a strong base.
A rule for where the double bond tend to form in elimination reactions. Zaitsev's rule states that a double bond will create the most stable alkene, the one where the carbon atoms that form the double bond hold the most substituents.
Add a catalytic amount of phosphoric acid with cyclohexanol in the first step. Heat this mixture, and upon heating phosphoric acid protonates the -OH group of cyclohexanol. Results into -OH⁺₂ which is a good leaving group. By the elimination of the -OH⁺₂ group, a carbocation generates, cyclohexene results in the last step by the elimination of the neighboring proton.
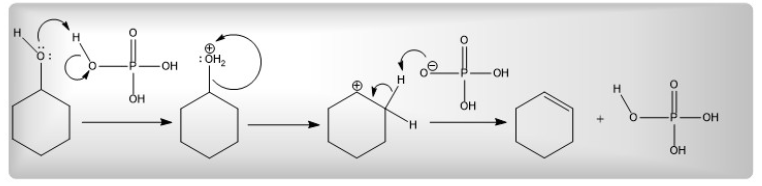
Figure 5. Elimination reaction of cyclohexanol to create cyclohexene (Image source generated by my self)
With technological advances, it is much easier to explain complex and challenging processes with the help of simulations. Now, you can simulate experiments without the need for any valuable equipment.
In this regard, you can take help from Labster’s virtual lab simulations. These simulations engage students through interactive learning scenarios. Students dive into a 3D world, where they visually learn and apply their concepts to solving real-life problems.

In Labster’s interactive Elimination Reaction: Use cyclohexanol to create polymers Virtual Lab, you will learn the ins and outs of Elimination Reactions in Organic Chemistry. You will get hands-on experience with some alkyl halides and discover how they go through the E1 and E2 eliminations.

Labster helps universities and high schools enhance student success in STEM.
Request DemoRequest a demo to discover how Labster helps high schools and universities enhance student success.
Request Demo