Heading 1
Heading 2
Heading 3
Heading 4
Heading 5
Heading 6
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat. Duis aute irure dolor in reprehenderit in voluptate velit esse cillum dolore eu fugiat nulla pariatur.
Block quote
Ordered list
- Item 1
- Item 2
- Item 3
Unordered list
- Item A
- Item B
- Item C
Bold text
Emphasis
Superscript
Subscript
About This Simulation
Build your own spectrophotometer to measure the absorbance of a reaction product on Mars!
Learning Objectives
- Summarize how a spectrophotometer is used to measure absorbance data
- Link the major components of a spectrophotometer to their functions within the instrument
- Choose a useful wavelength for measuring a compound of interest using absorbance spectrum data
- Suggest a protocol for measuring the absorbance of a given substance using a spectrophotometer
- Apply the Beer-Lambert equation to absorbance data to determine substance concentration
About This Simulation
Lab Techniques
- Spectrophotometry
Related Standards
- Support for spectrophotometer introductions
- Generic support for spectrophotometer introductions
- Generic support for spectrophotometer introductions
Learn More About This Simulation
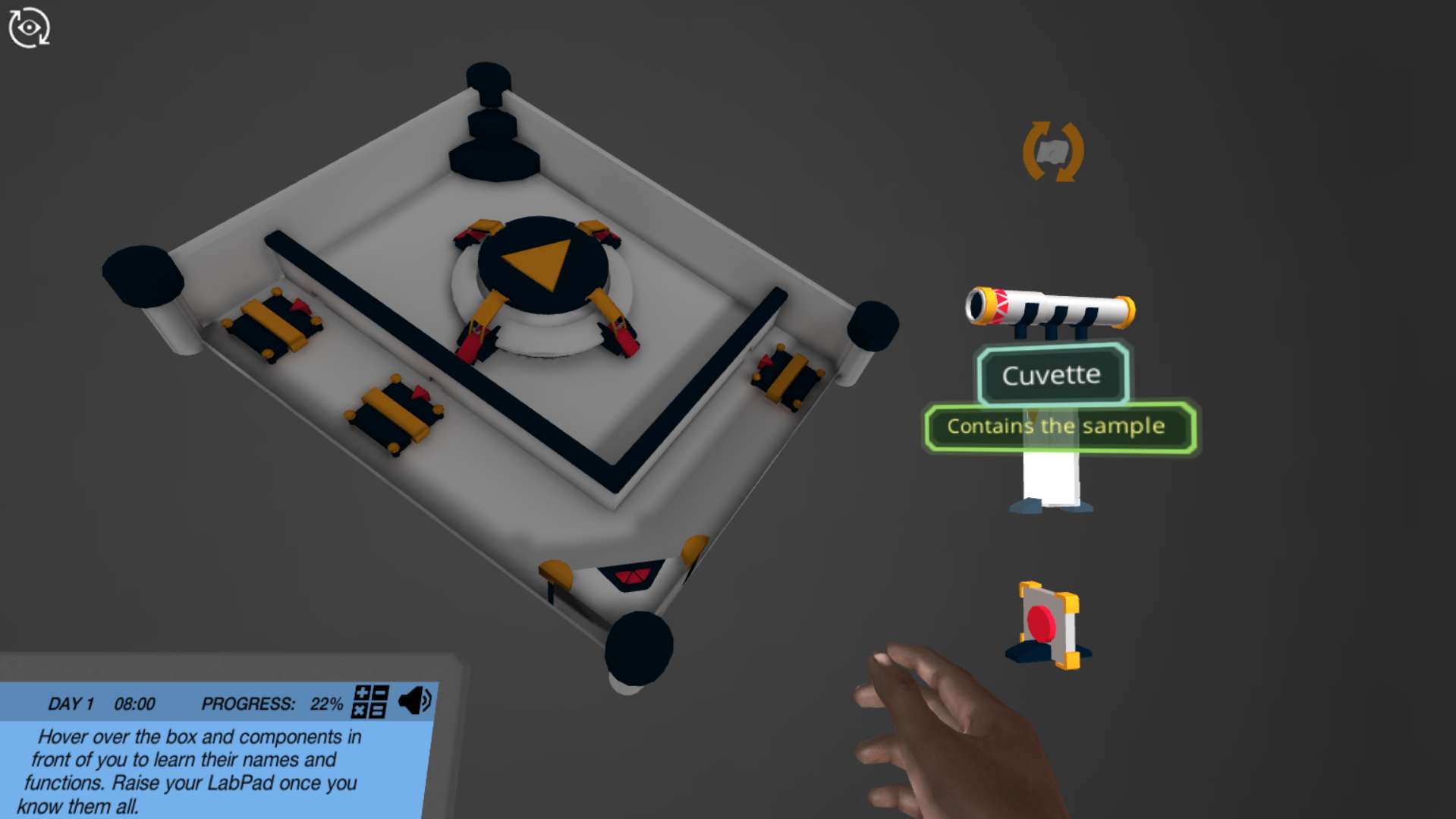
Determine a solution's concentration using light! In this simulation, you will build your own spectrophotometer - a machine that measures how much light is absorbed by a solution. Using the spectrophotometer and Beer-Lamberts law, you will be able to determine how much product is made after you add a new Martain catalyst to your reaction.
Build your spectrophotometer
Go under the hood of the spectrophotometer to understand how its components fit together. Try out different configurations of the components and see how they impact the light's path. The complexity of the spectrophotometer you build will increase over three levels going from a linear setup of three components, to a non-linear setup with six components. Can you figure out what each component does, and how they all work together in the finished instrument?
Operate a spectrophotometer and collect your own data
Once you’ve successfully built your instrument, it’s time to try it out! To get started, you’ll need to interpret spectra to determine the optimal wavelength your reaction product absorbs. Once you’re ready, set the baseline, and you’re off! Observe how the absorbance changes when you increase the reactant and catalyst concentration. Then link these changes in absorbance back to the amount of product and reaction rate.
Build confidence in applying Beer-Lambert Law to your data
Throughout the simulation, the different conceptual elements of Beer-Lambert law are explained as the spectrophotometer’s function is explored. Will you be able to rise to the final challenge, and convert your absorbance data into concentration data using Beer-Lambert's law?
For Science Programs Providing a Learning Advantage
Boost STEM Pass Rates
Boost Learning with Fun
75% of students show high engagement and improved grades with Labster
Discover Simulations That Match Your Syllabus
Easily bolster your learning objectives with relevant, interactive content
Place Students in the Shoes of Real Scientists
Practice a lab procedure or visualize theory through narrative-driven scenarios


FAQs
Find answers to frequently asked questions.
Heading 1
Heading 2
Heading 3
Heading 4
Heading 5
Heading 6
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat. Duis aute irure dolor in reprehenderit in voluptate velit esse cillum dolore eu fugiat nulla pariatur.
Block quote
Ordered list
- Item 1
- Item 2
- Item 3
Unordered list
- Item A
- Item B
- Item C
Bold text
Emphasis
Superscript
Subscript
A Labster virtual lab is an interactive, multimedia assignment that students access right from their computers. Many Labster virtual labs prepare students for success in college by introducing foundational knowledge using multimedia visualizations that make it easier to understand complex concepts. Other Labster virtual labs prepare learners for careers in STEM labs by giving them realistic practice on lab techniques and procedures.
Labster’s virtual lab simulations are created by scientists and designed to maximize engagement and interactivity. Unlike watching a video or reading a textbook, Labster virtual labs are interactive. To make progress, students must think critically and solve a real-world problem. We believe that learning by doing makes STEM stick.
Yes, Labster is compatible with all major LMS (Learning Management Systems) including Blackboard, Canvas, D2L, Moodle, and many others. Students can access Labster like any other assignment. If your institution does not choose an LMS integration, students will log into Labster’s Course Manager once they have an account created. Your institution will decide which is the best access method.
Labster is available for purchase by instructors, faculty, and administrators at education institutions. Purchasing our starter package, Labster Explorer, can be done using a credit card if you are located in the USA, Canada, or Mexico. If you are outside of North America or are choosing a higher plan, please speak with a Labster sales representative. Compare plans.
Labster supports a wide range of STEM courses at the high school, college, and university level across fields in biology, chemistry, physics, and health sciences. You can identify topics for your courses by searching our Content Catalog.