Heading 1
Heading 2
Heading 3
Heading 4
Heading 5
Heading 6
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat. Duis aute irure dolor in reprehenderit in voluptate velit esse cillum dolore eu fugiat nulla pariatur.
Block quote
Ordered list
- Item 1
- Item 2
- Item 3
Unordered list
- Item A
- Item B
- Item C
Bold text
Emphasis
Superscript
Subscript
Simulation Details
Learning Objectives
- Explain solid, liquid, and gas states in terms of particle interaction and bonding energy
- Describe and explain the characteristics of a phase change
- Read a phase diagram and explain each region
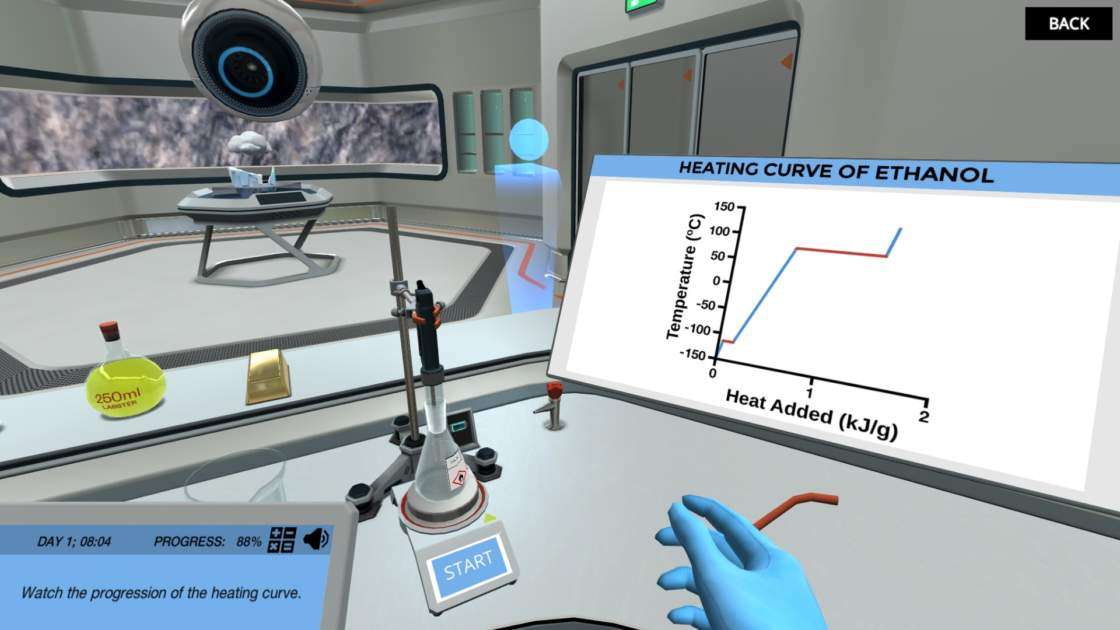
- Interpret a heating curve of a given substance
- Explain the difference between a heating curve and a phase diagram, and the difference between specific heat and latent heat
Lab Techniques
- Heating Curves
- Phase Changes
Related Standards
- Early Stage Bachelors Level
- US College Year 1
- US College Year 2
- FHEQ 6
- EHEA First Cycle
- EHEA Short Cycle
- HS-PS1-3
- HS-PS1-4
- HS-PS1-7
- HS-PS1-5
- PS1.A-E2
- Chemistry 2.2 Intermolecular Force and Potential Energy
- Chemistry 3.1. Intermolecular Forces
- Chemistry 3.2 Properties of Solids
- Chemistry 4.4 Physical and Chemical Changes
- Chemistry 6.5 Energy of Phase Changes
- Chemistry 4.4 Intermolecular forces
See How Science Programs Are Turning Curiosity Into Action and Mistakes Into Learning
Find Virtual Labs That Perfectly Match Your Syllabus
Get Instant, Personalized Recommendations to Help Reinforce Your Course Content

Frequently Asked Questions
Heading 1
Heading 2
Heading 3
Heading 4
Heading 5
Heading 6
Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat. Duis aute irure dolor in reprehenderit in voluptate velit esse cillum dolore eu fugiat nulla pariatur.
Block quote
Ordered list
- Item 1
- Item 2
- Item 3
Unordered list
- Item A
- Item B
- Item C
Bold text
Emphasis
Superscript
Subscript
A Labster virtual lab is an interactive, multimedia assignment that students access right from their computers. Many Labster virtual labs prepare students for success in college by introducing foundational knowledge using multimedia visualizations that make it easier to understand complex concepts. Other Labster virtual labs prepare learners for careers in STEM labs by giving them realistic practice on lab techniques and procedures.
Labster supports a wide range of STEM courses at the high school, college, and university level across fields in biology, chemistry, physics, and health sciences. You can identify topics for your courses by searching our Content Catalog.
Yes, Labster is compatible with all major LMS (Learning Management Systems) including Blackboard, Canvas, D2L, Moodle, and many others. Students can access Labster like any other assignment. If your institution does not choose an LMS integration, students will log into Labster’s Course Manager once they have an account created. Your institution will decide which is the best access method.